 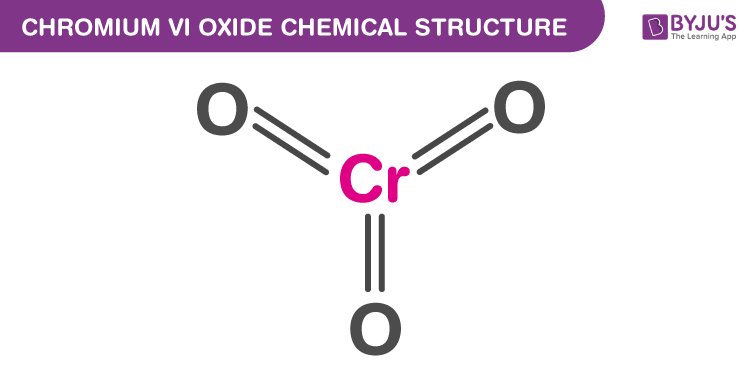
Ĭr(VI) enters many types of cells and under physiological conditions can be reduced by hydrogen peroxide (H2O2), glutathione (GSH) reductase, ascorbic acid, and GSH to produce reactive intermediates, including Cr(V), Cr(IV), thiylradicals, hydroxyl radicals, and ultimately, Cr(III). 
The balance that exists between extracellular Cr(VI) and intracellular Cr(III) is what ultimately dictates the amounts and rates at which Cr(VI) can enter cells and impart its toxic effects. If Cr(VI) is reduced to Cr(III) extracellularly, this form of the metal is not readily transported into cells and so toxicity is not observed. The reduction of Cr(VI) is considered to serve as a detoxification process when it occurs at a distance from the target site for toxic or genotoxic effect while reduction of Cr(VI) may serve to activate chromium toxicity if it takes place in or near the cell nucleus of target organs. It can be absorbed by the lung and gastrointestinal tract, and even to a certain extent by intact skin. In addition, although not approved for regulatory use, our recently developed method using suppressed conductivity, with detection limit of 1µg/L, eliminates the post-column derivatization step necessary for EPA 218.6 and 218.7 and may be used for screening and evaluation purposes for Cr(VI) contamination.Since Cr(III) is poorly absorbed by any route, the toxicity of chromium is mainly attributable to the Cr(VI) form. 
column can also be used in method 218.7 to get more sensitive results (AU179). 2mm i.d.), eluent flow rate, reagent coil volume, and reagent flow rate. The IC instrument combined with a guard column, to remove hydrophobic organics, and the analytical column can achieve the detection limit of 0.0044 or 0.0054µg/L, depending on the solid or liquid preservation reagent used.Īs shown in AU144 and AU179, multiple parameters can influence the detection limit: injection volume, column diameter (e.g. Our application updates AU144 and AU179 describe important methods developed for Cr(VI) analysis related to EPA Methods 218.6 and 218.7.ĮPA method 218.7 uses a Thermo Scientific ion chromatography (IC) system to measure chromium (VI), as described in Table 1 in the method. To comply with the new regulation, EPA method 218.7 must be used to achieve the low limit of detection. Prior to the new California regulation in 2014, EPA method 218.6 was approved for Cr(VI) analysis. For total chromium measurement in UCMR3, all the samples are required to be acid digested regardless of water turbidity. Nevertheless, all the samples need to be preserved in nitric acid to make sure the pH is less than 2 during storage. If you are measuring other water samplesĪcid digestion is only needed for total recoverable element analysis of water samples, with the exception of drinking water samples with turbidity less than 1 NTU.If the sample is drinking water with a turbidity of less than 1 NTU.If you are measuring the dissolved analytes or the total recoverable analytes.Only EPA 200.8, not 200.7, is approved for Unregulated Contaminant Monitoring Rule 3 (UCMR 3) for total chromium analysis because the method offers lower detection limit for the occurrence monitoring for the program.ĮPA method 200.8 can be used for metals analysis, including total chromium, in drinking water, surface water, groundwater, and wastewater. Total chromium in either drinking water or wastewater for regulatory compliance can be measured with inductively coupled plasma optical emission spectrometry (ICP-OES) using EPA method 200.7 or inductively coupled plasma mass spectrometry (ICP-MS) using EPA method 200.8. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
